2026 International Fall Summit
October 28, 2026 | 12:00 PM EST
MEDICALLY INTEGRATED PHARMACY ACCREDITATION
NCODA, a leading non-profit organization serving the patient-centered, medically integrated oncology community, offers the Center of Excellence Medically Integrated Pharmacy Accreditation Program. This pharmacy accreditation program helps pharmacies and healthcare organizations deliver high-quality, patient-centered care through leadership, clinical expertise, quality standards, and best practices. By earning accreditation, medically integrated pharmacies can demonstrate their commitment to excellence, strengthen pharmacy operations, and support better care for patients receiving cancer treatment.
Share

The NCODA Accreditation Program is based on compliance with the ASCO/NCODA Patient-Centered Standards for Medically Integrated Dispensing.
The ASCO/NCODA Quality Standards, published in the Journal of Clinical Oncology,
were developed to elevate oncology practice performance and promote fully medically integrated care. These standards support improved patient adherence while helping practices operate more efficiently and cost-effectively.
NCODA is committed to continuously advancing these standards. We collaborate with oncology practice experts to ensure they remain relevant, practical, and impactful for both patients and providers.
NCODA CoE MIP Program Highlights
- First and only accreditation program designed specifically for medically integrated pharmacies

- Built on innovative, patient-centered standards
- Minimizes administrative burden
- Improves patient outcomes while enhancing quality and reducing costs
- Strengthens medication adherence and reduces waste through cost avoidance
- Accelerates medication access and shortens fill times
- Enhances patient and clinician satisfaction
- Supports adoption of best practices and meaningful outcomes tracking
- Reduces clinical fragmentation through seamless coordination by the integrated care team
Pharmacy Accreditations
NCODA offers tailored pharmacy accreditation programs to meet the unique needs of practices, recognizing excellence in patient-centered care across diverse healthcare settings.
The NCODA Center of Excellence (COE) Medically Integrated Pharmacy
Accreditation Program, developed in collaboration with ASCO, recognizes practices that demonstrate excellence in delivering patient-centered, oncology-focused pharmacy care.
Canadian Pharmacy Accreditation Services (CPAS), an NCODA company, reflects NCODA’s expanding commitment to advancing patient-centered pharmacy care across the Canadian healthcare landscape. CPAS delivers a rigorous, yet accessible accreditation pathway designed for pharmacies of all sizes and specialties. Built on NCODA’s proven medically integrated pharmacy model, CPAS supports Canadian pharmacies in delivering the highest standards of clinical excellence, quality, and safety through a collaborative, patient-first approach.
NCODA Quality Standards
A comprehensive framework for Medically Integrated Dispensing (MID) services, focusing on patient-centered care, efficient workflows, data-driven decision-making, and Positive Quality Interventions (PQIs) that improve outcomes, enhance adherence, and streamline access to treatment.

Frequently Asked Questions
This section answers frequently asked questions about oncology pharmacy accreditation, including requirements, processes, and benefits. It’s designed for oncology pharmacists and healthcare professionals seeking to understand accreditation standards and how they support high-quality patient care.
The accreditation standards provide a framework to foster improvement in medication adherence, reduce waste due to cost avoidance, shorten medication fill times and improve patient and clinician satisfaction. Our accreditation program benefits Medically Integrated Pharmacies (MIPs) through the adoption of quality standards and best practices and tracking valuable patient outcomes. The NCODA CoE MIP accreditation is preferred for the Prime Therapeutics IntegratedRx Oncology program.
The NCODA CoE MIP Accreditation Program is designed to be sustainable, meaningful, and budget-friendly. Our focus is on patients and not the bottom line. Initial accreditation and subsequent reaccreditation range from $10,000 to $13,500.
NCODA provides comprehensive resources and handson support throughout the accreditation process — all designed not only to help practices achieve compliance, but to strengthen and enhance patient care. Practices have the option to purchase accreditation templates developed to support alignment with NCODA accreditation standards. These templates include, but are not limited to, clinical evaluation forms, error logs, new patient packets, staff training checklists, and Medically Integrated Pharmacy (MIP) standard operating procedures (SOPs). These structured tools guide practices step-bystep through the accreditation process while promoting standardized workflows, improved care coordination, and a consistent, high-quality patient experience.
Each practice is assigned a dedicated reviewer with expertise in pharmacy accreditation and pharmacy operations. This reviewer serves as a trusted resource throughout the entire process and is available by phone or email to answer questions, provide clarification, and assist with ensuring compliance. Their operational insight helps practices build efficient systems that support both regulatory standards and optimal patient outcomes. During the four-month self-study period, pharmacies work closely with the NCODA Accreditation Team to strengthen policies and procedures, bring documentation up to current standards, and determine an appropriate readiness date for the on-site review. The NCODA team provides ongoing, real-time feedback, allowing MIPs to confirm compliance before implementing new processes.
This proactive guidance not only minimizes noncompliant findings in the final self-study review report but also helps practices establish sustainable processes that enhance patient safety, improve medication management, and elevate the overall quality of care.
To apply for NCODA Center of Excellence MIP Accreditation, an organization must meet the following eligibility requirements:
- All members of the pharmacy care team are required to obtain NCODA membership within ninety (90) days of submitting the accreditation application.
- The organization must be a medically integrated pharmacy currently dispensing oral oncolytics to approximately 50% of its pharmacy patient population.
NCODA membership provides oncology care teams and healthcare professionals with access to specialized clinical resources, continuing education, and a international peer network. Members benefit from tools and guidance that support medically integrated dispensing, strengthen compliance with industry standards, and improve practice performance. Through collaboration, education, and shared best practices, NCODA helps enhance the quality and consistency of patient-centered cancer care.
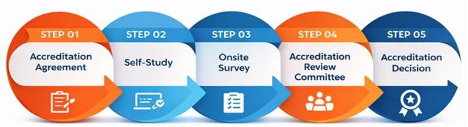
The accreditation process consists of five stages:
agreement, self-study, onsite review, accreditation review committee evaluation and final decision. The full process typically takes between 8 and 12 months to complete.
NCODA provides comprehensive training, guidance, and resources to support practices throughout the accreditation process. Accreditation training includes an overview of standards, criteria, and best practices, along with strategies for achieving a successful outcome.
Each practice is assigned a dedicated accreditation team, including a reviewer who provides documentation feedback and guidance, and a surveyor who conducts the onsite survey and completes the final report. This team offers ongoing support, standard interpretation, and resources throughout the 8–12 month accreditation process and beyond. In addition, members have access to a robust library of tools and resources designed to support patient management, financial assistance, cost avoidance, waste tracking, and staff development.






